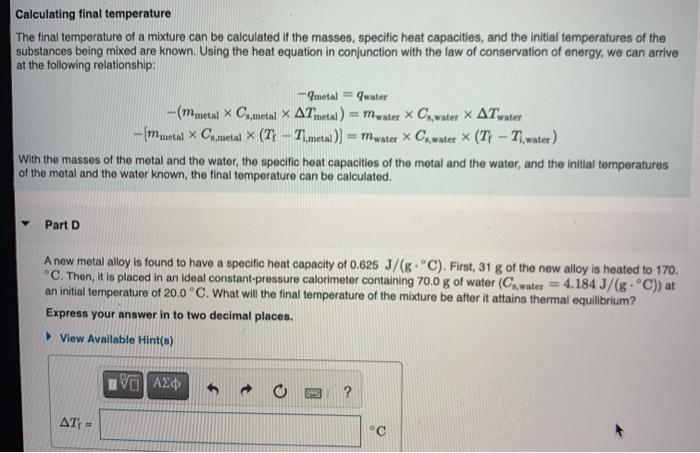
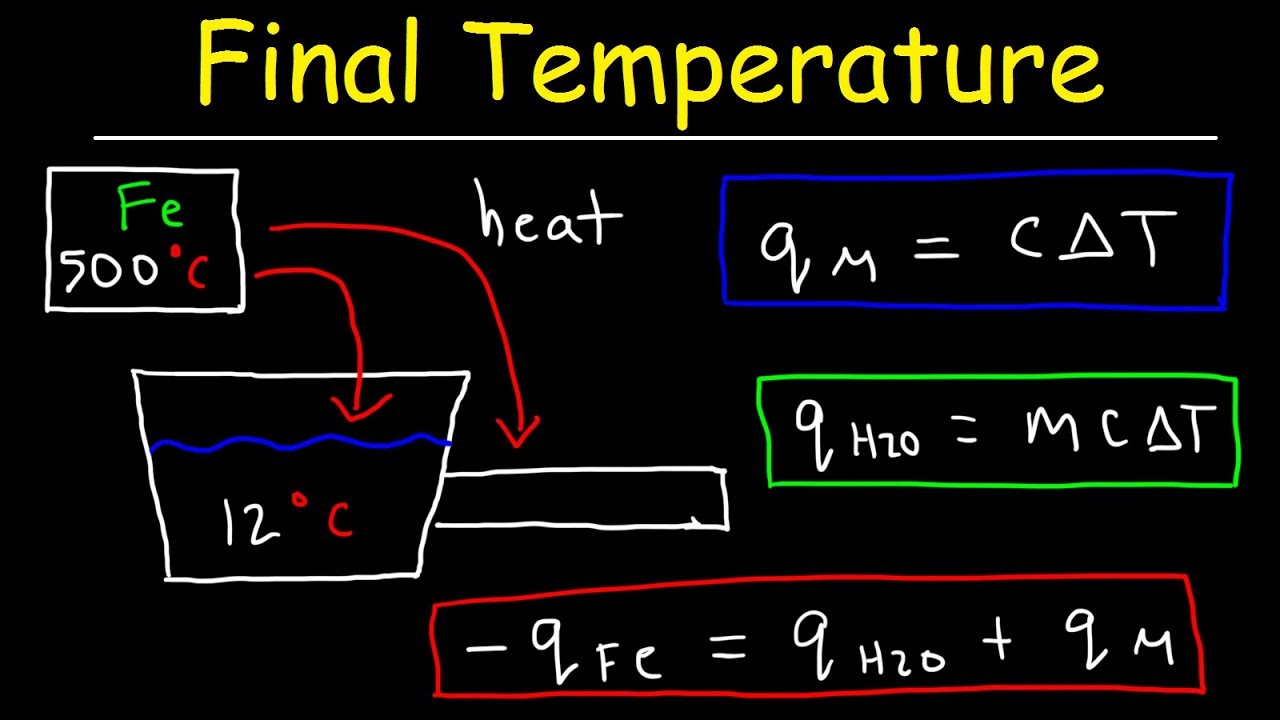
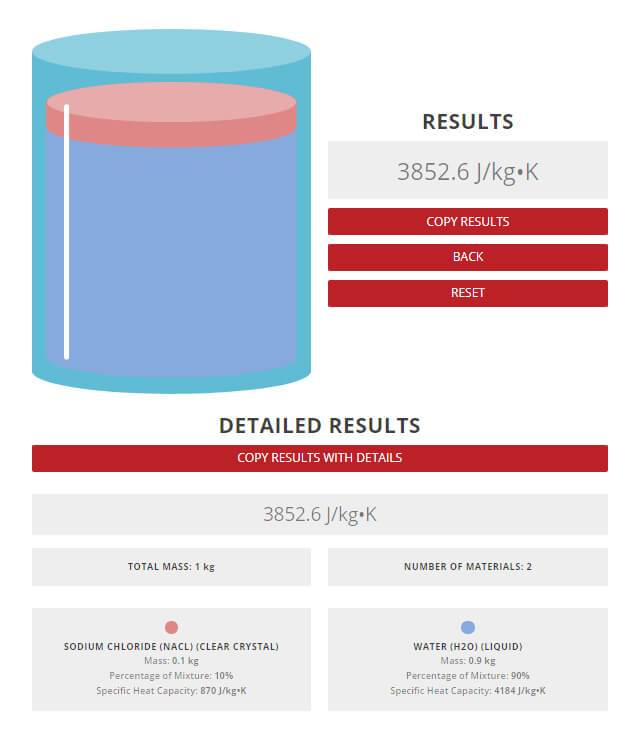
Find the specific heat capacities cv and cp for a gaseous mixture consisting of 7.0 g of nitrogen and 20 g of argon. - Sarthaks eConnect | Largest Online Education Community

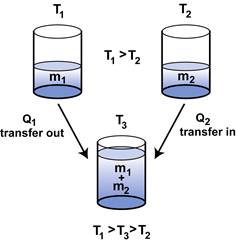
To measure the specific latent heat of ice by the method of mixtures Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online

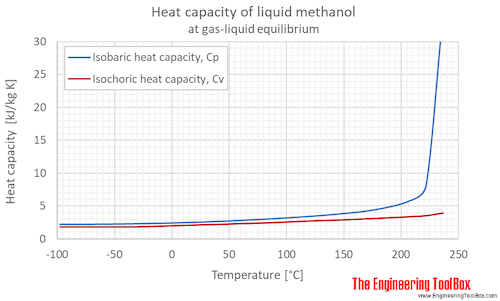
Thermal fluids with high specific heat capacity through reversible Diels-Alder reactions - ScienceDirect

What is Specific Heat Capacity? | How to Find Specific Heat Capacity - Video & Lesson Transcript | Study.com

SOLVED: 2 HCI(aq) Ba(OH)2(aq) > BaCl2(aq) +2H2o(U) AH–118 kJ Calculate the heat when 124.8mL of 0.5O0M HCl is mixed with 300.0 mL of 0.540M Ba(OHJ2 Assuming that the temperature of both solutions

SOLVED: A5. 50-g sample of aluminum pellets (specific heat capacity 0.89 J/'C g) and a 19.00-g sample of iron pellets (specific heat capacity 0.45 J/PC*g) are heated to 100.0 C. The mixture